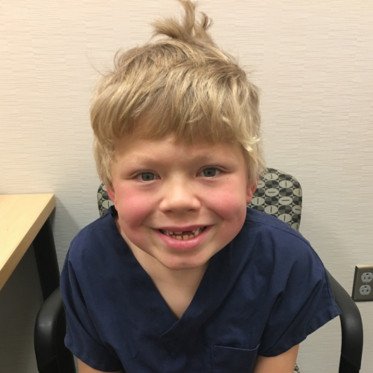
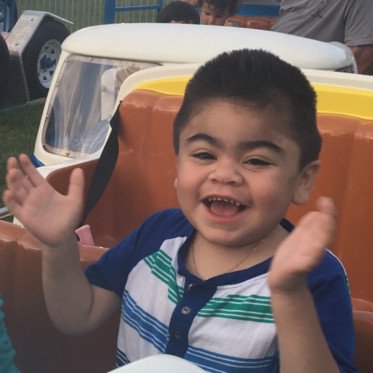
About Us
We are Project Alive
We exist to find a cure for Hunter syndrome through research and advocacy, and providing education and resources to the Hunter syndrome community. We are here to give a voice to the families and children touched by Hunter syndrome.

OUR PROMISE TO THE COMMUNITY
We Are Here For You
We welcome and embrace those families who are newly diagnosed. We hold close those families who are journeying alongside us. And we support and comfort those families whose children have passed – never forgetting the impact of their lives and the joy they brought us.
HERE FOR YOU
Meet the Project Alive Team
Get to know the people behind our amazing team at Project Alive and the people that support our cause.

See What We've Been Up To
Updates and News
October 26, 2024
WonderFall Family Festival
Project Alive and The Hunter Syndrome Foundation are teaming up to host the WonderFall Family Festival on October 26, 2024 at the Rough Rider's Stadium in Frisco, TX! This fall festival will be an inclusive, family friendly event to raise awareness about Hunter syndrome and support the work being done in research, advocacy, and community support by Project Alive.